About Himantura uarnak (Gmelin, 1789)
Scientific name: Himantura uarnak (Gmelin, 1789)
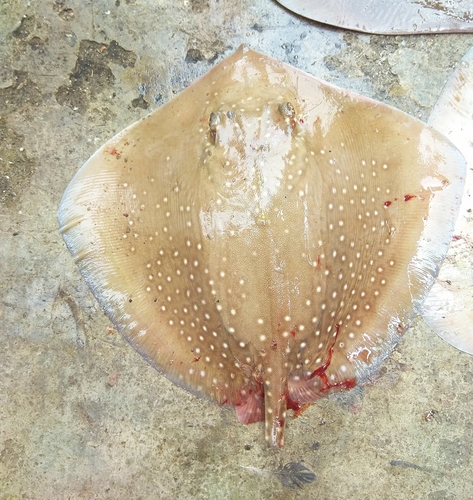
Description The reticulate whipray has a diamond-shaped pectoral fin disc that is wider than it is long, with nearly straight leading margins, and quadrangular snout and outer corners. The apex of the pectoral fin is narrowly rounded, and the anterior margin is almost straight. In juvenile individuals, the disc is roughly as wide as it is long, with a blunter snout and more rounded corners. Its eyes are small, with a small distance between the two eyes. Paired respiratory openings called spiracles sit immediately behind the eyes. Between the long, thin nostrils, there is a short, wide curtain of skin with a finely fringed rear margin. The mouth is relatively small, with a deep hollow at the center of the lower jaw and shallow grooves at the corners that extend onto the lower jaw. A row of 4–5 nipple-like papillae runs across the floor of the mouth. There are 26–40 upper tooth rows and 27–44 lower tooth rows. The pelvic fins are small and triangular. The extremely thin, whip-like tail measures 3–3.5 times the length of the disc when intact, and has no fin folds. Usually one serrated stinging spine sits on the upper surface of the tail, at some distance from the base. Adult rays have a wide band of flattened, heart-shaped dermal denticles that stretches from between the eyes to the tail spine; this band increases in density as the ray ages. Adults also have two large pearl thorns at the center of the back. The section of the tail behind the spine is covered in small thorns. The dorsal denticle band is mostly fully developed by the time juveniles reach 50 cm (20 in) across the disc. The reticulate whipray's coloration changes a great deal based on age and location. Adults typically have a dorsal pattern of many closely spaced dark brown spots or reticulations over a beige to yellow-brown background. Past the stinging spine, the color becomes blackish, with lighter bands along the sides. This textured color pattern is often called a "beehive pattern," and it is a prominent example of a natural Voronoi cell, which is a structure made of repeated arrays of polygons. This pattern is considered an evolutionary optimization that minimizes the energy required to form it, while also providing cryptic coloration. The ventral side is pale white and has no markings. Juveniles are yellowish on the upper side, with tiny, densely packed dark spots. There are around seven spots in a line between the spiracles, and three rows of spots in front of the sting. Juvenile spots are generally larger; spots decrease in size as the ray ages to form the distinct honeycomb/leopard pattern, an example of negative allometric growth. This is a large species, with reported maximums of 2 m (6.6 ft) across the disc, 6 m (20 ft) in total length, and 120 kg (260 lb) in weight.
Distribution and habitat The reticulate whipray lives in coral reefs, brackish water, and marine environments. Within these habitats, it is found inshore and on soft substrates. It occurs in northern Taiwan, the Western Indian Ocean (including Malaysia, Myanmar, and the broader Indo-Pacific), the Red Sea, west as far as Natal in South Africa, and the Arabian Sea. Thirty years ago, it entered the Mediterranean Sea via the Suez Canal, and is now common in the southeastern Mediterranean. H. australis, a recently described species that was formerly mistaken for H. uarnak, appears to be common only in Australian waters, where it ranges from Shark Bay to Brisbane. The reticulate whipray is bottom-dwelling, and is most often found over sandy flats near beaches, in lagoons, and around coral reefs, from the intertidal zone to offshore waters 50 m (160 ft) deep or more. It shifts use of microhabitats (intertidal sand flats, shallow subtidal sand flats, and seagrass beds) between warm and cold seasons. In Shark Bay, it uses intertidal sand flats during the warm season, and moves to slightly deeper seagrass patches in the cold season. This species tolerates low salinities, and has been recorded entering estuaries and mangrove swamps. However, reports of it entering fresh water in Southeast Asia are unverified and may be misidentifications. Its preferred water temperature range is 23–26 °C (73–79 °F).
Biology and ecology During the day, the reticulate whipray is generally inactive and spends most of its time resting motionless on the seafloor, sometimes buried in sand. In Shark Bay, Western Australia, this ray can be found resting alone or in small groups in very shallow water during high tide. The ray's evolution in shallow, bright light conditions has led to vision specialized for photopic vision. Its eyes have high concentrations of ganglion cells and cones, plus spatial resolving power that suggests color vision, as well as elongated horizontal specialization for visual acuity. A lateral line runs all the way to the tip of its extremely long tail, which gives the ray advance warning of approaching predators such as Indo-Pacific bottlenose dolphins (Tursiops aduncus) and hammerhead sharks (Sphyrna). A very small number of rare encounters have recorded dolphins being injured or even killed by wounds from a reticulate whipray's barb lodged in the dolphin's side. These encounters are theorized to most often happen in shallow shore feeding sites where this species tends to live. The severity of reaction to a barb wound is not correlated with the age or size of the whipray. As whiprays age, they are more likely to lose the venom glands and the protective sheath of their stinging spine. The cowtail stingray (Pastinachus sephen) prefers to rest alongside the reticulate whipray rather than alongside other members of its own species, because the reticulate whipray's longer tail provides better predator detection. These mixed-species groups often arrange themselves into a "rosette" with their tails pointing radially outward, to maximize predator awareness. The reticulate whipray preys on a wide range of benthic and neritic organisms, including crabs, shrimps, mantis shrimps, bivalves, gastropods, worms, jellyfish, and bony fishes. In the western Indian Ocean, roughly two-thirds of its diet is made up of fish, particularly ponyfish and anchovies, with shrimps and other crustaceans making up most of the remaining portion. By contrast, reticulate whiprays in Australian waters are apparently not piscivorous, and are known to eat penaeid prawns. H. uarnak fills an important ecological role as a mesopredator, and is thought to have a strong top-down impact on nearshore environments when human disturbance is absent. Overfishing of tertiary predators (mainly sharks) leads to higher population densities of benthic feeding predators like the reticulate whipray, which is associated with trophic cascades. Known parasites of this species include Anthrobothrium loculatum, Dendromonocotyle colorni, Halysiorhynchus macrocephalus, Monocotyle helicophallus, M. multiparous, M. spiremae, Thaumatocotyle australensis, and Tylocephalum chiralensis. The marine bacterium Vibrio alginolyticus has been recorded infecting reticulate whiprays, causing lethargy, whitish necrotic skin lesions, and skin ulcers. This infection can be treated with amikacin to prevent death. Like other stingrays, the reticulate whipray is aplacental viviparous: developing embryos are first nourished by yolk, which is later replaced by histotroph ("uterine milk," enriched with proteins and lipids) produced by the mother. Females give birth to up to five pups in summer, after a one-year gestation period. Off the coast of South Africa, newborn rays measure 28–30 cm (11–12 in) across the disc, and reach sexual maturity at a disc width of about 1 m (3.3 ft), which corresponds to an age of 4 to 5 years. Off Australia, newborns measure 21–28 cm (8.3–11.0 in) across the disc, and males mature at 82–84 cm (32–33 in) across the disc. Juveniles of H. leoparda and H. undulata differ in birth size, disc shape, denticle development, and spotting amount, and are more distinct from each other than adult Himantura species are. Shark Bay may act as a nursery area for young reticulate whiprays. While species in the 'uarnak' species complex look and function similarly, interspecific hybridization has not yet been clearly confirmed by nuclear genetic markers. It is possible that interspecific differences in the female urogenital sinus, plus differences in the distal length and shape of the male clasper, create reproductive isolation between species.